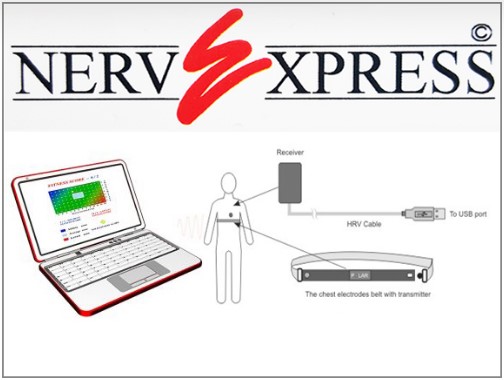
NerveExpress
Oorspronkelijke prijs was: €2,850.00.€1,200.00Huidige prijs is: €1,200.00. excl. btw
Productomschrijving
Intellewave HRV system is a fully automated cardiac monitoring device that provides quantitative assessment of Heart Rate Variability analysis. IntelleWave testing allows physicians to monitor physiological activity and assess the state of a patient’s autonomic function through analysis of Spectral Function Components of R-R intervals Variability. HRV testing with IntelleWave system enables physicians to detect specific types of autonomic dysfunction that may be linked to a multitude of clinical diagnoses. Additionally, IntelleWave is a tool for overall, general assessment of a patient’s physiology, and as such, provides a comprehensive, in-depth patient evaluation, often missing in standard medical practice. Such an ability to assess one’s physiology determines the wide range of IntelleWave’s clinical applications and distinguishes it favorably from all other diagnostic devices based on HRV analysis. In the medical field IntelleWave system can be used by health-care providers to objectively confirm or exclude cases of Cardiovascular Autonomic Neuropathy (CAN), Diabetic Autonomic Neuropathy (DAN) and other diseases or malfunctions of the Autonomic Nervous System (ANS). For instance, with the information provided by IntelleWave clinicians are able to recognize cases of beta-blocker overdose or risk of sudden cardiac arrest. Moreover, IntelleWave can monitor ANS activity by providing up to 24 hours of “realtime” quantitative assessment of spectral function components of HRV (HF and LF). Note: all clinical correlations of HRV analysis data are performed solely by physician. IntelleWave system’s proprietary algorithm (US Patent 2012018916) uses novel artificial intelligence techniques to differentiate between high frequency (HF) and low- frequency (LF) components of spectral function. This unique representation of test results allows physicians to recognize up to 81 different variations of the relationship between LF and HF components.